A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. A ten-fold serial dilution could be 1 M, 0.1 M, 0.01 M, 0.001 M ... Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in concentration curves with a logarithmic scale. A tenfold dilution for each step is called a logarithmic dilution or log-dilution, a 3.16-fold (100.5-fold) dilution is called a half-logarithmic dilution or half-log dilution, and a 1.78-fold (100.25-fold) dilution is called a quarter-logarithmic dilution or quarter-log dilution. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
In biology and medicine[edit]
In biology and medicine, besides the more conventional uses described above, serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample. As, for instance, the number and size of bacterial colonies that grow on an agar plate in a given time is concentration-dependent, and since many other diagnostic techniques involve physically counting the number of micro-organisms or cells on specials printed with grids (for comparing concentrations of two organisms or cell types in the sample) or wells of a given volume (for absolute concentrations), dilution can be useful for getting more manageable results.[1] Serial dilution is also a cheaper and simpler method for preparing cultures from a single cell than optical tweezers and micromanipulators.[2]
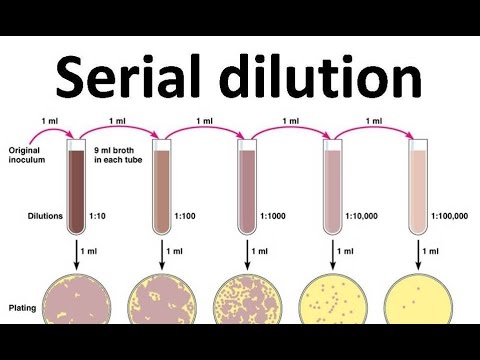
Serial Dilution Many procedures performed in modern biology and chemistry laboratories require sets of solutions that cover a range of concentration. s. These include quantifying the number of bacteria in a sample using plate counts and development of standard curves for quantitative colorimetric, radiometric and enzymatic assays.
Serial Dilution Many procedures performed in modern biology and chemistry laboratories require sets of solutions that cover a range of concentration. s. These include quantifying the number of bacteria in a sample using plate counts and development of standard curves for quantitative colorimetric, radiometric and enzymatic assays. Serial Dilution Of Soil Sample Procedure DOWNLOAD (Mirror #1) b89f1c4981 The most commonly used method for soil sampling would be based on soil types.This video describes how to set up a serial dilution of bacteria from a soil sample.Serial dilution and plating of a bacterial culture. Do your best to devise a serial dilution procedure. 4/5/2005 Lab 2. Perform a serial dilution, which are a series of dilutions, when the final volume is a large value like 10,000 mL, for example. In this case, make a 1 mL to 100 mL dilution first and from that solution take another 1 mL into another 100 mL. The final solution is a 1 to 10,000 mL (100 mL x 100 mL) dilution. A serial dilution is a series of sequential dilutions used to reduce a dense culture of cells to a more usable concentration. The easiest method is to make a series of 1 in 10 dilutions. In this method, exactly 1 ml of each successive dilution is transferred into exactly 9 ml of liquid in a dilution blank, creating a 1/10 dilution. A serial dilution is any dilution in which the concentration decreases by the same factor in each successive step. In serial dilutions, you multiply the dilution factors for each step. The dilution factor or the dilution is the initial volume divided by the final volume.
In homeopathy[edit]
Serial dilution is one of the core foundational practices of homeopathy, with 'succussion', or shaking, occurring between each dilution. In homeopathy, serial dilutions (called potentisation) are often taken so far that by the time the last dilution is completed, no molecules of the original substance are likely to remain.[3][4]
See also[edit]
References[edit]
- ^K. R. Aneja. Experiments in Microbiology, Plant Pathology and Biotechnology. New Age Publishers, 2005, p. 69. ISBN81-224-1494-X
- ^Booth, C.; et al. (2006). Extremophiles. Methods in microbiology 35. Academic Press. p. 543. ISBN978-0-12-521536-7.
- ^Weissmann, Gerald (2006). 'Homeopathy: Holmes, Hogwarts, and the Prince of Wales'. The FASEB Journal. 20 (11): 1755–1758. doi:10.1096/fj.06-0901ufm. PMID16940145. Retrieved 2008-02-01.
- ^Ernst, Edzard (November 2005). 'Is homeopathy a clinically valuable approach?'. Trends in Pharmacological Sciences. 26 (11): 547–548. CiteSeerX10.1.1.385.5505. doi:10.1016/j.tips.2005.09.003. PMID16165225.
- Michael L. Bishop, Edward P. Fody, Larry E. Schoeff. Clinical Chemistry: Principles, Procedures, Correlations. Lippincott Williams & Wilkins, 2004, p. 24. ISBN0-7817-4611-6.
External links[edit]
Serial Dilution Procedure For Soil Sample
- How to Make Simple Solutions and Dilutions, Bates College

Retrieved from 'https://en.wikipedia.org/w/index.php?title=Serial_dilution&oldid=904833671'
•••brightly colored science test tubes image by Steve Johnson from Fotolia.com
By Contributor
Learning how to perform a dilution correctly in chemistry or microbiology class will improve your laboratory techniques and ensure results that are more accurate. These science classes require different dilution techniques and not everyone knows a difference exists. Use these dilution methods during your next laboratory experiment and watch your yields or counts improve.
- Label dilutions properly for easy identification Do no replace liquid lost during mixing. This additional volume of liquid reduces the accuracy of the dilution.
- Always add acid to water. Adding water to acid results in violent reactions and can cause serious injury. Use a pipette bulb and stay away from the older practice of mouth pipetting.
Use volumetric glassware when doing dilutions for chemistry or analytical methods. Serological pipettes and graduated cylinders are fine for microbiology dilutions used to reduce micro levels to a countable number.
Begin with a liquid stock solution. This may be a straight liquid sample or a solution made from a powder or liquid, diluted to a known volume.
Make a chemistry or analytical dilution by taking a volumetric amount of a solution, using a volumetric pipette, into a volumetric flask of the desired final volume. For example, a 1 to 100 dilution in chemistry requires the use of a 1.0 mL volumetric pipette and a 100 mL volumetric flask. The final volume of the dilution will be 100 mL (1 mL of stock solution plus 99 mL of diluent, the solution used for diluting).
Perform a microbiology dilution by taking a serological pipette and measuring a volume of the stock solution into a beaker. Then add the diluent using a graduated cylinder and mix in the beaker. A 1 to 100 dilution in microbiology requires the addition of 1 mL of stock solution to 100 mL of diluent for a final volume of 101 mL.
Use the proper diluent identified in the method for the dilution. Liquids such as media, buffer and water are common microbiology diluents. Chemistry methods will specify diluents like solvents, acids, bases and water.
Swirl the flask halfway through the dilution to mix. Then continue to add the remaining solution.
Use a dropper for adding the final amounts of diluent in small drops for an accurate final volume measurement.
Read the final volume by looking at the meniscus. View the meniscus by holding up the flask or beaker to eye level. The shape seen at the top of the liquid level that looks like a smile or upside-down umbrella is the meniscus. The bottom, not the sides that stretch up the sides of the glass, of the meniscus in the center should line up with the line drawn on the flask for an accurate measurement.
Add a magnetic stir bar to the final dilution and place on a stir plate to mix. Alternatively, stopper the flask and swirl, then hold the stopper on with the thumb and flip the flask upside down and back several times to mix.
Perform a serial dilution, which are a series of dilutions, when the final volume is a large value like 10,000 mL, for example. In this case, make a 1 mL to 100 mL dilution first and from that solution take another 1 mL into another 100 mL. The final solution is a 1 to 10,000 mL (100 mL x 100 mL) dilution.
Perform acid dilutions differently by adding a little water in the flask before adding the smaller volume of acid. Dilute to volume as normally required.
Serial Dilution Procedure Pdf
- brightly colored science test tubes image by Steve Johnson from Fotolia.com
